In this article 
View / Download
pdf version of this article
Over the past several years, the worldwide market for generic drugs has grown at a faster rate than the pharmaceutical
industry as a whole. This is largely due to strong efforts to contain drug costs by government agencies, as well as growing
confidence in generic quality by both health professionals and consumers.1
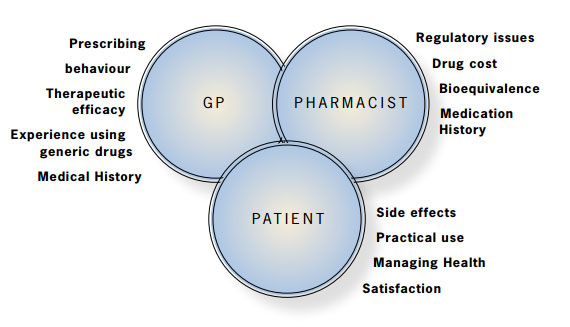
When an innovator product is replaced with a generic alternative, there are many issues that must be taken into consideration.
Physician attitude towards generic substitution is most often related to their general prescribing behaviour, perception
of therapeutic efficacy, beliefs about generic drugs and previous experience with using generic alternatives, including
any negative effects.2
Pharmacists may consider regulatory issues, drug class, cost and bioequivalency information when dispensing generic
products as well as a patient’s medical and medication history and their comfort with a brand change.1
The main issues confronting patients undergoing a brand change to a generic substitute include overall satisfaction,
changes in therapeutic effects, side effects, practical use (e.g. size, shape, appearance), willingness to take medication
and concern about managing their health condition.3 Increasing age is associated with less
favourable attitude towards generic drugs and patients are often less accepting of using a generic drug to treat a serious
disease.4
Patients who received information from their physician or pharmacist about generic substitution were more likely
to have switched from the branded product.
SSRI antidepressants and brand changing
The issues surrounding brand changing may become more complex when the drug in question is used to treat a serious medical
condition such as depression. Consideration must be given to the perceptions and attitudes of a patient receiving antidepressant
therapy and how this may affect the way they accept a change to their drug therapy, both physiologically and psychologically.
Patients are often concerned that a change in their antidepressant medication may result in reduced clinical effect and/or
increased side effects.
The generic form of paroxetine due to be released in New Zealand (Loxamine) has met Medsafe’s bioequivalence standards,
in accordance with international guidelines (see bioequivalence article). This means that
the risk of a reduced response, increased side effects or the appearance of discontinuation syndrome due to a significant
change in the dose received is very unlikely. It is probable that perceptions and attitudes to the change may be stronger
determinants of response and acceptance of change than bioequivalence issues.
Brand changing with other SSRIs has occurred in New Zealand. In the case of citalopram the generic was associated with
a significant number of reports to MARC. Most of these related to a perceived loss or change in therapeutic effect but
it has not been determined if this was due to reduced drug content or attitudes and perceptions.
The change to Loxamine will involve a large number of people using a drug which has not been used before in New Zealand.
This drug is however, already widely available throughout Europe, with sales for 2005 and 2006 in excess of 150 million
tablets (Personal communication - Regulatory Affairs Manager, Pacific Pharmaceuticals). No patterns of adverse effects
have been reported with Loxamine, indicating that problems in New Zealand will be unlikely. However pharmacists are in
an ideal position to monitor patient feedback, response, side effects and attitudes to the change.
Although therapeutic problems were sometimes the most important adversity, more commonly it was a failure of communication
which contributed largely to the lack of satisfaction.
Satisfaction with the communication received was closely correlated with satisfaction about the change to the generic
drug itself.
To report any suspected adverse reactions, contact
Centre for Adverse Reactions Monitoring - CARM
Phone 03 479-7247, http://carm.otago.ac.nz
PHARMAC help line 0800 66 00 50
Due to the nature of mental illness and/or previous experiences, there may be some groups of people that are less accepting
of a change to a generic drug. These people may require extra information or counselling through the process and include;
- People with recurring episodes of depression who responded well when previously treated with the innovator product
- People who have been stabilised on an innovator product for a long time
- Subgroups of people with OCD, anxiety disorders or co-morbid psychiatric disorders
- People who have been poorly informed about a previous brand change
- People who had a negative experience with a previous brand change
How do patients perceive the risk of changing to a generic drug?
One of the main barriers in changing brands of medication is the patient's perception of risk.
A study conducted among American consumers of health care services found that between 14% and 54% believed that generic
prescription drugs are both less safe and less effective than the innovator product. Perception of risk was dependent
on the severity of the medical condition being treated, with patients with a heart condition perceiving the highest risk.
5 Another
study found significant differences in acceptability rates of generic substitution according to pharmacological class
of drug - generic substitution was less accepted for drugs acting on the CNS (including antidepressants).6
Although generic drugs are clinically equivalent to their innovator counterparts, there will always remain a population
of people who believe they are receiving an inferior product and therefore will be dissatisfied with the change. A recent
study conducted in Norway assessed patient attitudes towards generic drug substitution.
- 36% reported one or more negative experiences in relation to the substitution
- 21% reported an overall negative experience after the change
- 12% reported experiencing side effects
- 18% felt that the generic drug had a weaker effect than the innovator medication
- 41% would not change if they had no economic incentive to do so
- 27% said that they would never accept substitution
There were no actual reports of clinical failure of the generic drugs. Negative experiences with generic drug substitution
were not related to polypharmacy, patient age or gender.3
At the time of brand changing, generic drug substitution for many patients is not considered an equal alternative to
innovator drugs. However, over time most patients do accept the change and the generic drug attains the status of the
branded version. A study in the United States has found that since 1979, 94 to 97% of patients in the state of New Jersey
have agreed to use a generic substitute.1
Differences in appearance and brand loyalty
Generic drugs often differ in appearance and packaging from the innovator product which may cause anxiety and confusion
in patients. Those receiving drug treatment for psychological disorders may be especially vulnerable to this.5 Changing
the colour, taste or form of a drug can result in non-compliance.
Although there is no evidence that generic drugs are inferior to innovator products patients may often resist changing
from a brand they know well to a generic equivalent which may look different.6 Patients
prefer to stick to a brand name drug if they have already been familiarised with it.1 The
longer a generic brand has been available, the more likely it is used by pharmacists and prescribers. Prescriber and pharmacist
habits and preferences may be influenced by informational constraints, loyalties to drug companies and desire to satisfy
patients.4
Individual educational intervention that lasted less than five minutes in most cases, resulted in a high rate of
generic acceptability.
Patient information programmes make brand changing easier
The Norwegian study found that patients who received information from their physician or pharmacist about generic
substitution were more likely to have changed from the innovator product.
Over half of the patients (54%) remembered receiving information from their pharmacist on generic substitution and 24%
remembered receiving this information from their doctor.3
A recent study based in Spain assessed acceptance of substitution of innovator drugs for generic drugs for chronic conditions
in primary care. Of the patients who received verbal information and written material on generic drugs, 98.9% agreed to
receive a generic formulation. The reasons for refusal in the remaining patients included the influence of physicians
other than the general practitioner, patients’ satisfaction with the innovator product and adverse effects.
There was no statistically significant difference between patients that agreed and those that didn’t agree with substitution
based on age, gender or educational level. There were however, significant differences in acceptability rates according
to individual primary care centres, suggesting differences in quality of information provided. It was concluded that individual
educational intervention (that lasted less than five minutes in most cases) in patients with repeat prescriptions resulted
in a high rate of generic acceptability. All patients accepted the intervention. The intervention also helped to stimulate
health practitioner’s knowledge of generic drugs.6
In a study that assessed the impact of introducing generic substitutes to patients in a general practice clinic in Scotland,
70% accepted the generics and were satisfied. Of the remaining patients, 20% were still taking the branded drug, 4% were
on other prescribed treatment, 4% had stopped treatment and 3% were purchasing their own alternative. Patients were either
sent an explanatory letter detailing the change or were informed when first collecting their repeat prescription. Reasons
for dissatisfaction were largely due to the quality of information provided to the patient rather than problems with the
generic drug itself. Almost three quarters of patients (73%) could recall being informed of the change in at least one
way. Satisfaction with the communication received was closely correlated with satisfaction about the change to the generic
drug itself. After four months, generic prescribing increased from 37% to 58%.7
The results of these studies suggest that appropriate care must be taken to inform patients properly. Interviews with
patients highlighted the aspects of the experience which caused most dissatisfaction. Although therapeutic problems were
sometimes the most important adversity, more commonly it was a failure of communication which contributed largely to the
lack of satisfaction. Patients were much more likely to be willing to try the new treatment if they thought they understood
the reason for the change. Almost all patients were aware that saving money was at least part of the reason for the brand
change and they all felt that a trial of a cheaper alternative was reasonable.7
Dissatisfaction usually centres around two main issues:
Power; patients feel weak or not in control of their health management
if they perceive a change has been forced upon them.7
Communication; patients require empathy from their healthcare provider
in order to feel satisfied, so they will accept major change if it is delivered in a manner which makes them feel valued.
This also helps to avoid much of the negative feeling generated by the change itself and enhances the patient/healthcare
provider relationship which can easily be damaged by these changes.7
References
- Suh D. Trends of generic substitution in community pharmacies. Pharm World Sci 1999;21(6):260-65.
- Banahan B, Kolassa E. A physician survey on generic drugs and substitution of critical dose medications. Arch Intern
Med 1997;157(18):2080-88.
- Kjoenniksen I, Lindbaek M, Granas A. Patients’ attitudes towards and experiences of generic drug substitution in
Norway. Pharm World Sci 2006;28:284 - 289.
- Mott D, Cline R. Exploring generic drug use behaviour: The role of prescribers and pharmacists in the opportunity
for generic drug use and generic substitution. Med Care 2002;40(8):662-74.
- Ganther J, Kreling D. Consumer perceptions of risk and required cost savings for generic prescription drugs. J Am
Pharm Assoc 2000;40:378-83.
- Valles J, Barreiro M, Cereza G, Ferro J, Martinez M, Escriba J, et al. A prospective multicenter study of the effect
of patient education on acceptibility of generic prescribing in general practice. Health Policy 2003;65:269-75.
- Dowell J, Snadden D, Dunbar J. Changing to generic formulary: how one fundholding practice reduced prescribing costs.
BMJ 1995;310:505-508.